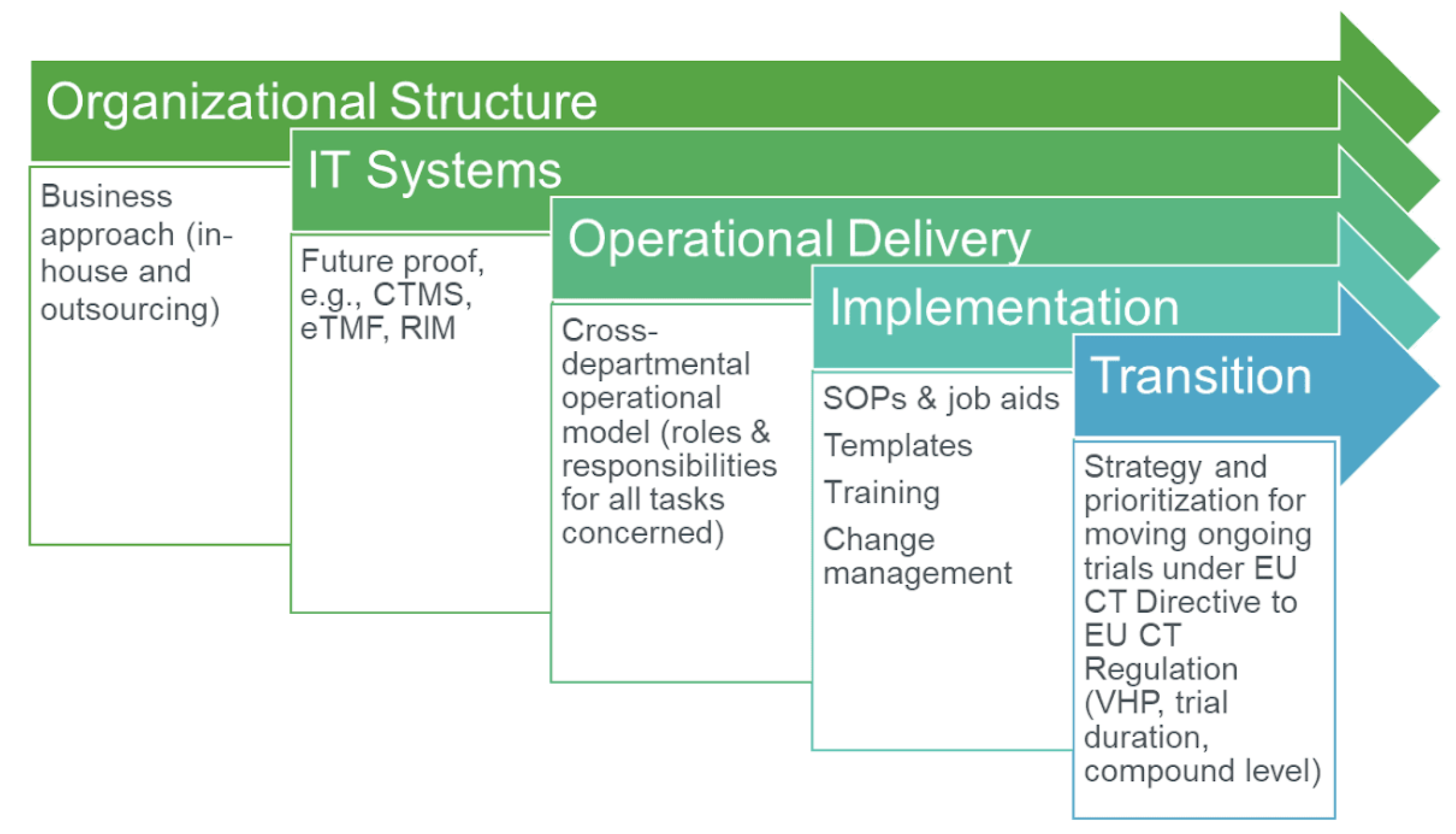
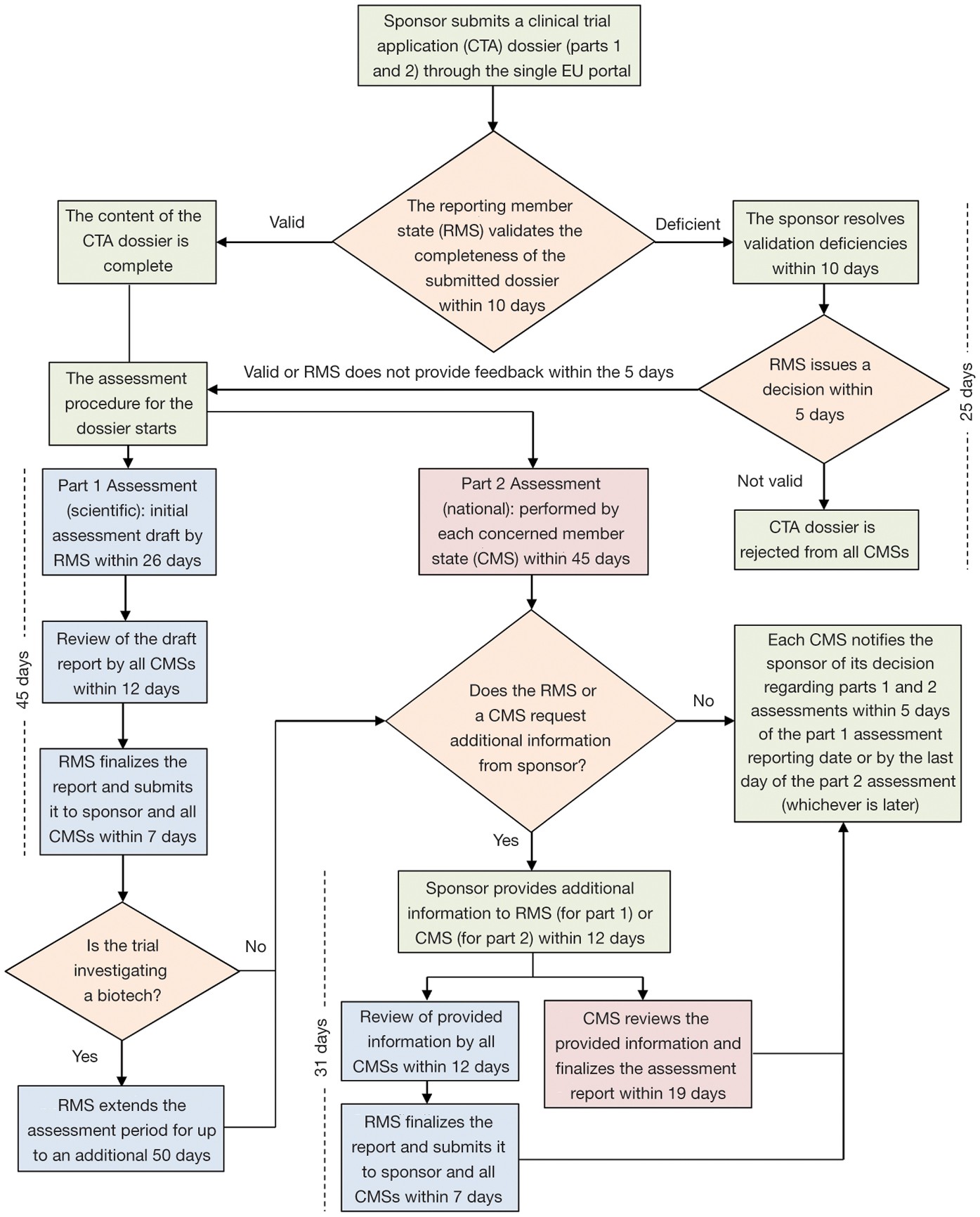
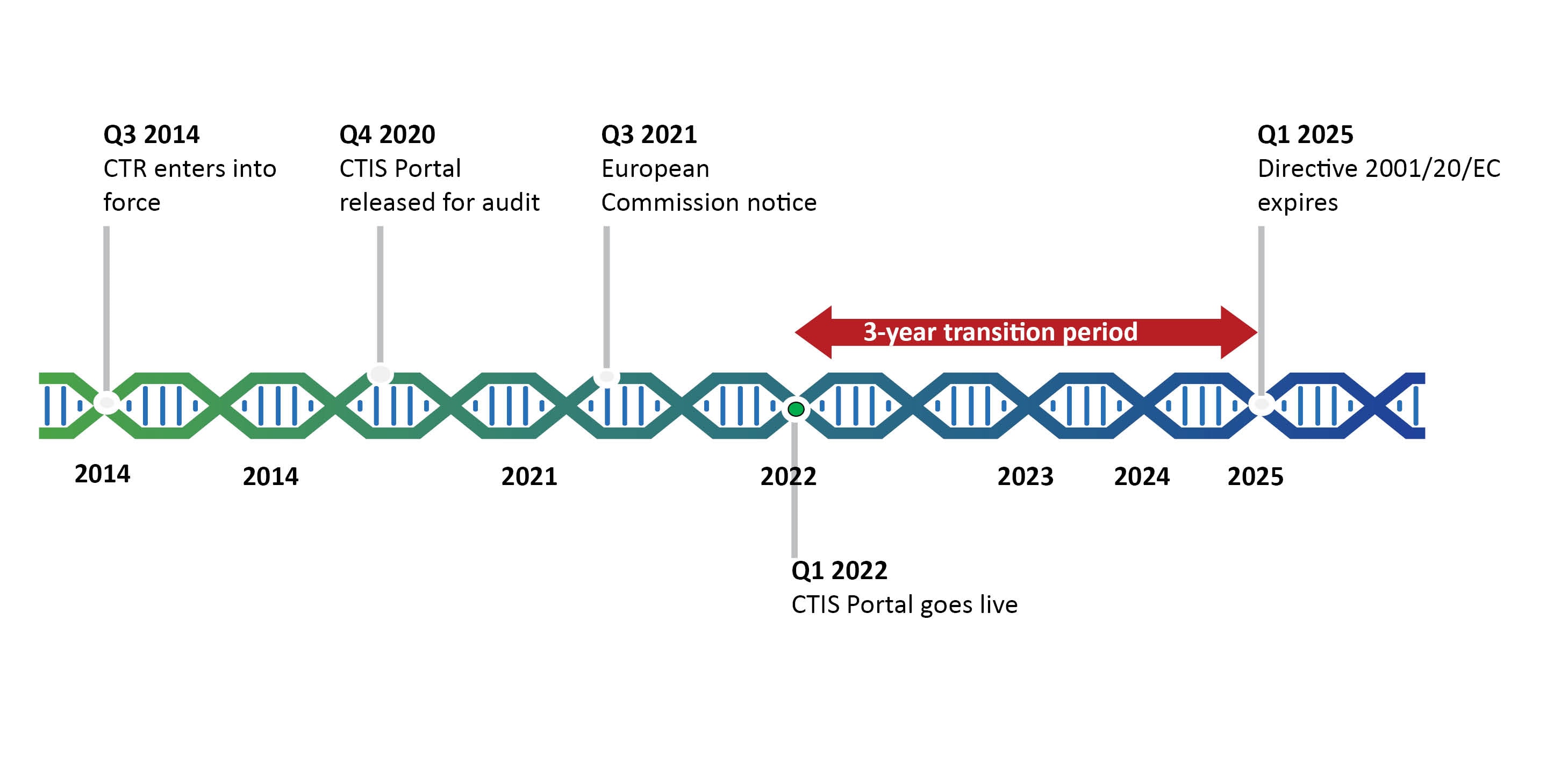
EUROPEAN COMMISSION Brussels, 11/04/2012 sanco.ddg1.d.6(2012)501417 VOLUME 10 - G Date of discussion of draft by the ad-hoc gro

EORTC EU Clinical Trials Directives Organisation and Implementation of Cancer Clinical Trials Anastassia Negrouk EORTC Regulatory Affairs Manager Intergroup. - ppt download

Exploring the Impact of the New European Directive on the Pharmaceutical Industry - Clinical Trials Arena

Will the EU Clinical Trials Regulation Support the Innovative Industry in Bringing New Medicines Faster to Patients? | SpringerLink

Directive 2001/20/EC : Clinical trials on medicinal products for human use - Free PDF download | M A N O X B L O G

When innovation outpaces regulations: The legal challenges for direct‐to‐patient supply of investigational medicinal products - Malone - 2022 - British Journal of Clinical Pharmacology - Wiley Online Library