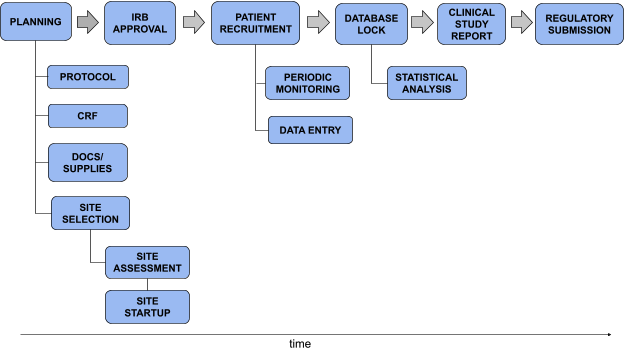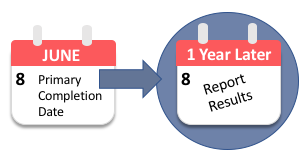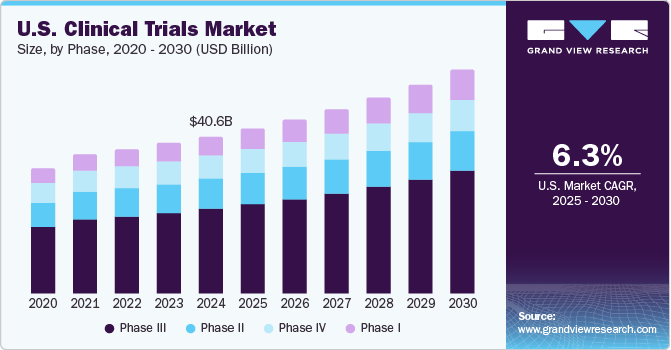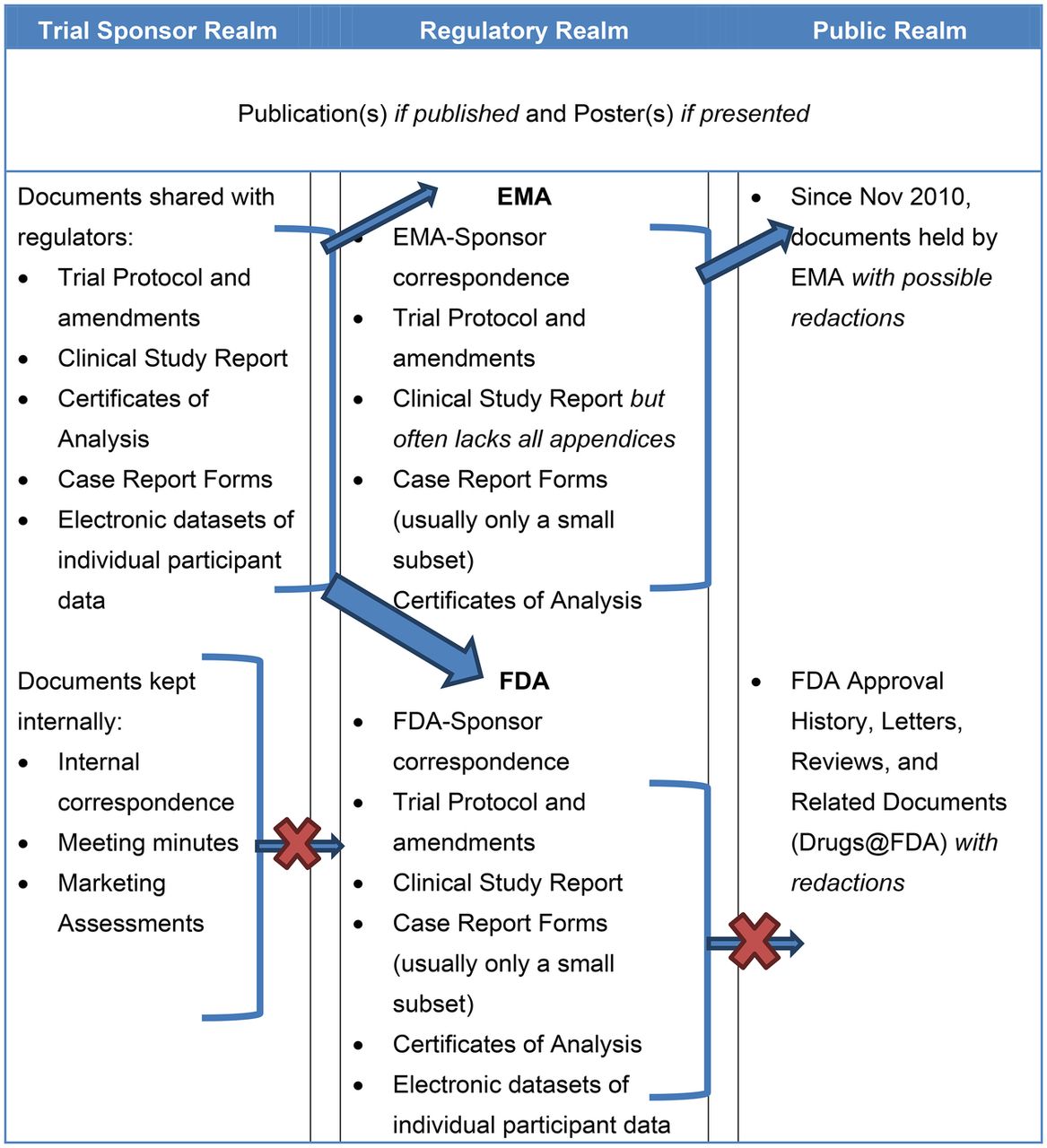
Clinical study reports of randomised controlled trials: an exploratory review of previously confidential industry reports | BMJ Open

Clinical Trial Report Template (6) - TEMPLATES EXAMPLE | TEMPLATES EXAMPLE | Report template, Clinical trials, Template google
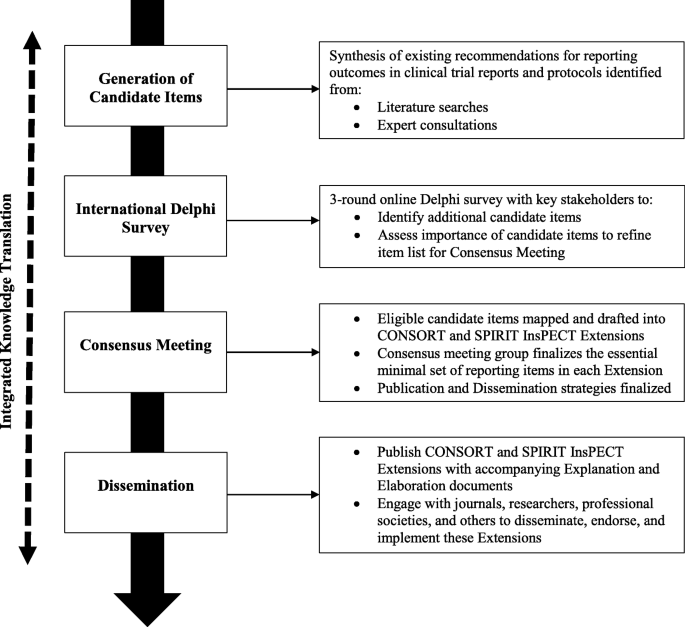
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Example of Clinical Trial Assessment of Infrastructure Matrix (CT AIM)... | Download Scientific Diagram
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine

Will clinical trial data disclosure reduce incentives to develop new uses of drugs? | Nature Biotechnology

Appendix F Illustrative Data Fields for the Results Summary | Developing a National Registry of Pharmacologic and Biologic Clinical Trials: Workshop Report | The National Academies Press