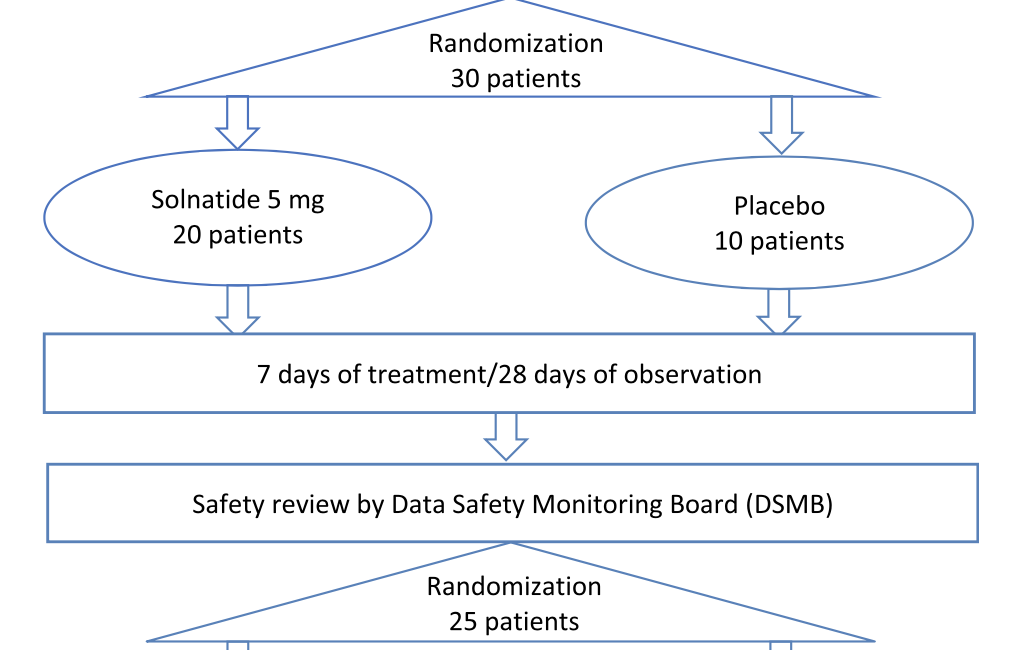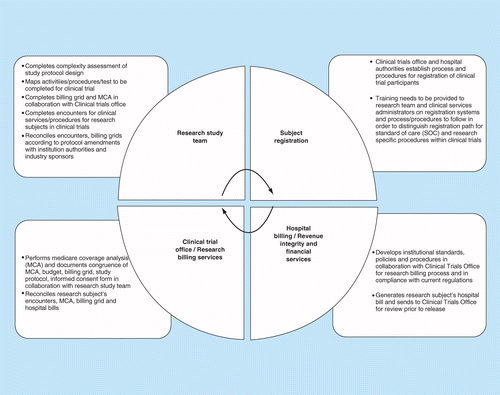
Optimization of protocol design: a path to efficient, lower cost clinical trial execution | Future Science OA
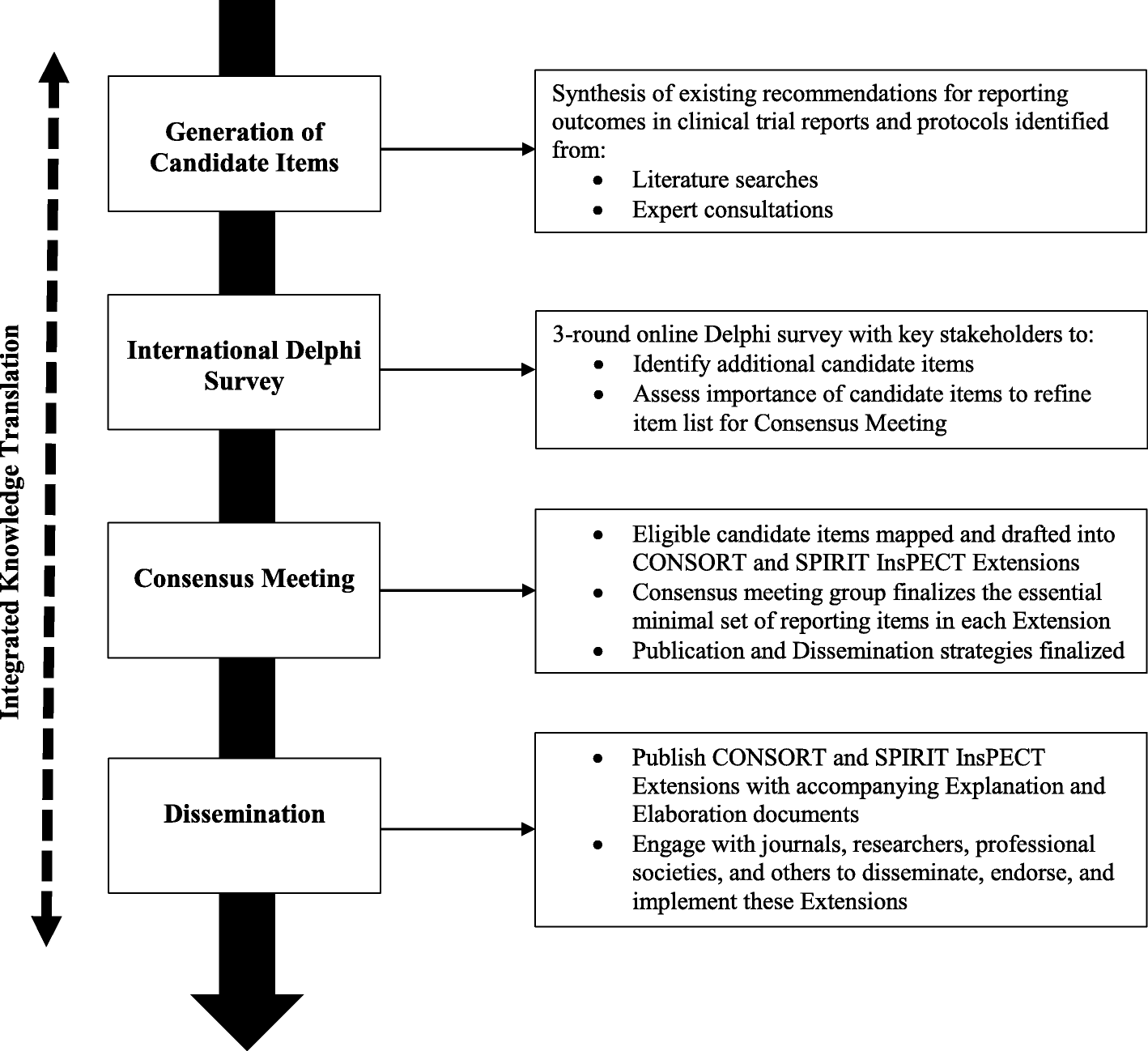
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text
An interactive retrieval system for clinical trial studies with context-dependent protocol elements | PLOS ONE

Clinical Trial Documentatino | Clinical trials study, Clinical trials, Contract research organization

PPT – Clinical trial protocol writing: Challenges and Guidelines PowerPoint presentation | free to download - id: 906c6c-ODNhN
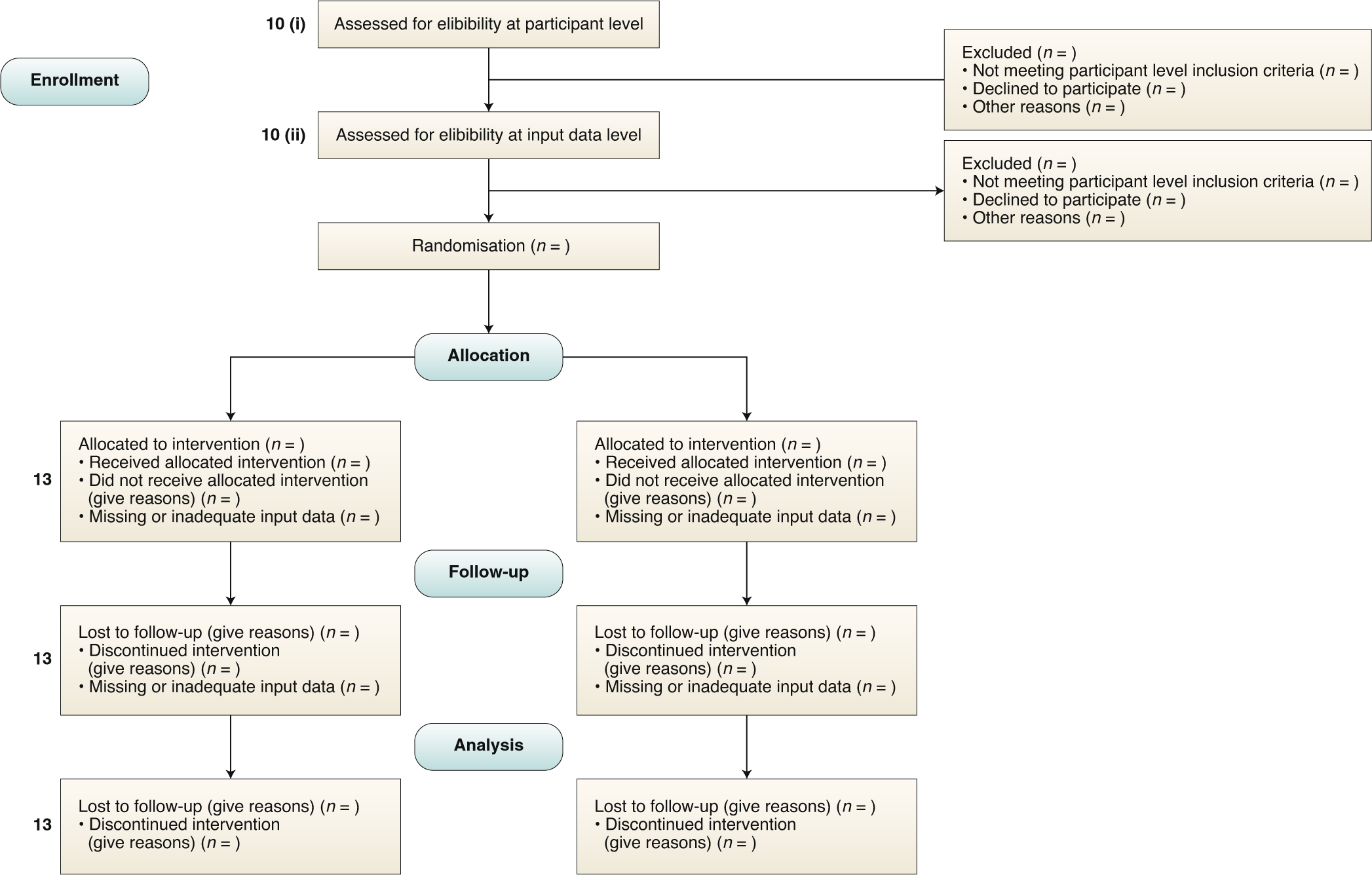
Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension | Nature Medicine

Clinical trial protocol components onboarding mobile app page screen with concepts 2283568 Vector Art at Vecteezy
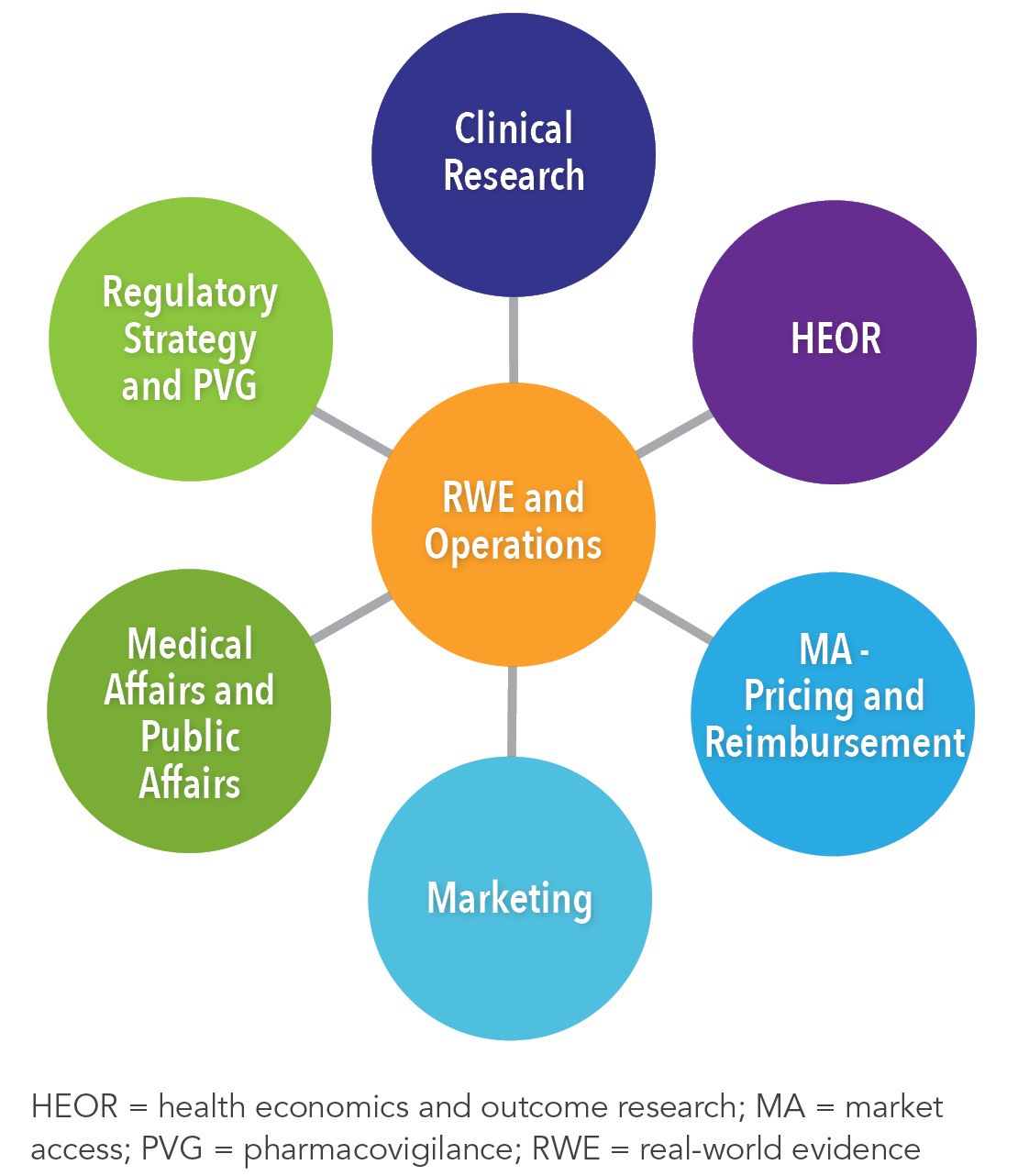
White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera